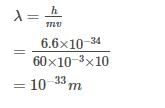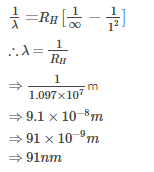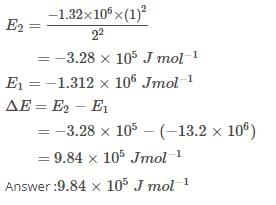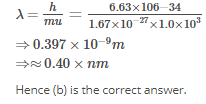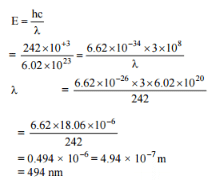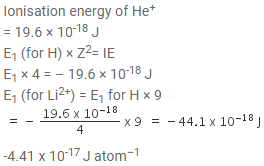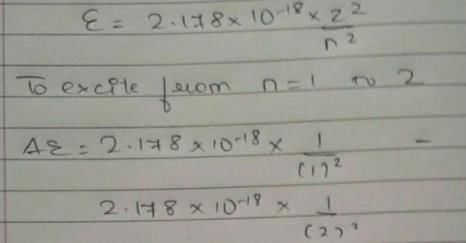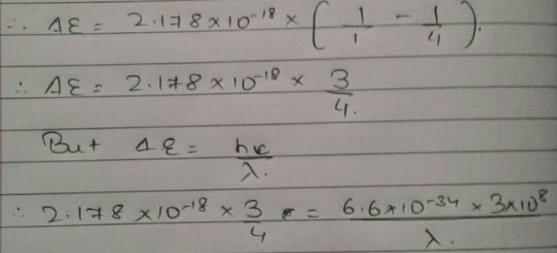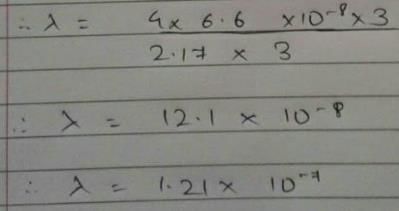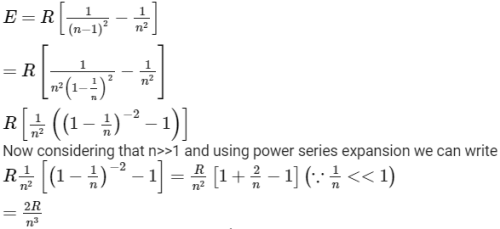JEE Previous Year Questions- Structure of Atom with solutions - Free MCQ
MCQ Practice Test & Solutions: Test: JEE Previous Year Questions- Structure of Atom (28 Questions)
You can prepare effectively for JEE Chapter-wise Tests for JEE Main & Advanced with this dedicated MCQ Practice Test (available with solutions) on the important topic of "Test: JEE Previous Year Questions- Structure of Atom". These 28 questions have been designed by the experts with the latest curriculum of JEE 2026, to help you master the concept.
Test Highlights:
- - Format: Multiple Choice Questions (MCQ)
- - Duration: 28 minutes
- - Number of Questions: 28
Sign up on EduRev for free to attempt this test and track your preparation progress.
An atom has a mass of 0.02 kg & uncertainity in its velocity is 9.218 × 10-6 m/s then uncertainity in position is (h = 6.626 × 10-34 J - s) [AIEEE- 2002]
Detailed Solution: Question 1
Energy of H–atom in the ground state is -13.6 eV , Hence energy in the second excited state is–
Detailed Solution: Question 2
Uncertainty in position of a particle of 25 g in space is 10-5 m. Hence uncertainty in velocity (ms-1) is (Planck's constant h = 6.6 × 10-34 Js) [AIEEE- 2002]
Detailed Solution: Question 3
The orbital angular momentum for an electron revolving in an orbit is given by 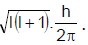 . This momentus for an s-electron will be given by - [AIEEE- 2003]
. This momentus for an s-electron will be given by - [AIEEE- 2003]
Detailed Solution: Question 4
In Bohr series of lines of hydrogen spectrum, third line from the red end corresponds to where one of the following inter-orbit jumps of electron for Bohr orbits in an atom of hydrogen. [AIEEE- 2003]
Detailed Solution: Question 5
The de Broglie wavelength of a tennis ball mass 60 g moving with a velocity of 10 mt. per second is approximately - [AIEEE- 2003]
Detailed Solution: Question 6
Which of the following sets of quantum numbers is correct for an electron in 4f orbital ? [AIEEE- 2004]
Detailed Solution: Question 7
Consider the ground state of Cr atom (Z = 24). The numbers of electrons with the azimuthal quantum numbers, l = 1 and 2 are, respectively [AIEEE- 2004]
Detailed Solution: Question 8
The wavelength of the radiation emitted, when in a hydrogen atom electron falls from infinity to stationary state 1, would be (Rydberg constant = 1.097×107 m-1) [AIEEE- 2004]
Detailed Solution: Question 9
Which one of the following sets of ions represents the collection of isoelectronic species ? [AIEEE- 2004]
Detailed Solution: Question 10
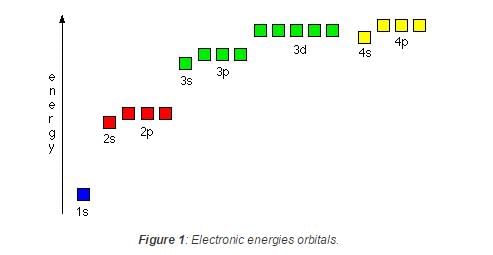
In a multi-electron atom, which of the following orbitals described by the three quantum members will have the same energy in the absence of magnetic and electric fields ? [AIEEE- 2005]
(a) n = 1, l = 0, m = 0
(b) n = 2, l = 0, m = 0
(c) n = 2, l = 1, m = 1
(d) n = 3, l = 2, m = 1
(e) n = 3, l = 2, m = 0
Detailed Solution: Question 11
Of the following sets which one does NOT contain isoelectronic species ? [AIEEE- 2005]
Detailed Solution: Question 12
According to Bohr's theory, the angular momentum of an electron in 5th orbit is - [AIEEE 2006]
Detailed Solution: Question 13
Uncertainty in the position of an electron (mass = 9.1 × 10-31 kg) moving with a velocity 300 m/s, accurate upto 0.001 %, will be (h = 6.63 × 10-34 Js) [AIEEE 2006]
Detailed Solution: Question 14
Which of the following sets of quantum numbers represents the highest energy of an atom ? [AIEEE 2007]
Detailed Solution: Question 15
The ionization enthalpy of hydrogen atom is 1.312 × 106 J mol-1. The energy required to excite the electron in the atom from n = 1 to n = 2 is [AIEEE 2008]
Detailed Solution: Question 16
In an atom, an electron is moving with a speed of 600 m/s with an accuracy of 0.005%. Certainity with which the position of the electron can be located is (h = 6.6×10-34 kg m2s-1 ,mass of electron, em = 9.1×10-31 kg ) [AIEEE 2009]
Detailed Solution: Question 17
Calculate the wavelength (in nanometer) associated with a proton moving at 1.0×103ms-1
(Mass of proton = 1.67×10-27 kg and h = 6.63×10-34 Js ) : [AIEEE 2009]
Detailed Solution: Question 18
The energy required to break one mole of Cl - Cl bonds in Cl2 is 242 kJ mol-1. The longest wavelength of light capable of breaking a single Cl - Cl bond is(c = 3 x 108 ms-1 and NA = 6.02 x 1023 mol-1) [AIEEE 2010]
Detailed Solution: Question 19
Ionisation energy of He+ is 19.6 x 10-18 J atom-1. The energy of the first stationary state (n = 1) of Li2+ is
[AIEEE 2010]
Detailed Solution: Question 20
Iron exhibits +2 and +3 oxidation states. Which of the following statements about iron is incorrect ?
[AIEEE 2012]
Detailed Solution: Question 21
The electrons identified by quantum numbers 'n' and l : [AIEEE 2012]
(a) n = 4, l = 1
(b) n = 4, l = 0
(c) n = 3, l = 2
(d) n = 3, l = 1
can be placed in order of increasing energy as :
Detailed Solution: Question 22
The standard reduction potentials for Zn2+/Zn, Ni2+/Ni, and Fe2+/Fe are -0.76, -0.23 and -0.44 V respectively. The reaction X + Y2+ → X2+ + Y will be spontaneous when: [AIEEE 2012]
Detailed Solution: Question 23
The first ionisation potential of Na is 5.1 eV. The value of electron gain enthalpy of Na+ will be:
[AIEEE 2013]
Detailed Solution: Question 24
Energy of an electron is given by E = -2.178 × 10-18 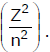 Wavelength of light required to excite an electron in an hydrogen atom from level n = 1 to n = 2 will be (h = 6.62 × 10-34 Js and c = 3.0 × 108 ms-1)
Wavelength of light required to excite an electron in an hydrogen atom from level n = 1 to n = 2 will be (h = 6.62 × 10-34 Js and c = 3.0 × 108 ms-1)
[AIEEE 2013]
Detailed Solution: Question 25
Which of the following is the wrong statement ? [AIEEE 2013]
Detailed Solution: Question 26
Which of the following arrangements does not represent the correct order of the property stated against it?
[AIEEE 2013]
Detailed Solution: Question 27
In a hydrogen like atom electron make transition from an energy level with quantum number n to another with quantum number (n - 1). If n >> 1, the frequency of radiation emitted it proportional to [AIEEE 2013]
Detailed Solution: Question 28
446 docs|929 tests |